25/02/2021
Published in
The Conversation
Ignacio López-Goñi
Full Professor of Microbiology of the University of Navarra
 A year ago I wrote a article graduate the same way Ten good news about coronavirus. The goal was to show that science, knowledge and cooperation are essential to fight the pandemic. We do not know what will happen in the coming months and new genetic variants are a cause for uncertainty, but one year later the message is the same: advances in science encourage us to be optimistic and to see "the bottle as half full".
A year ago I wrote a article graduate the same way Ten good news about coronavirus. The goal was to show that science, knowledge and cooperation are essential to fight the pandemic. We do not know what will happen in the coming months and new genetic variants are a cause for uncertainty, but one year later the message is the same: advances in science encourage us to be optimistic and to see "the bottle as half full".
1. There are more articles on SARS-CoV-2 and COVID-19 than on malaria.
A year ago we were amazed that in little more than a month after the first cases were reported there were already more than 164 scientific articles in PubMed on the new virus and the disease. Now that figure has multiplied by more than 600, exceeding 100,000 articles, more than those appearing under the heading of "malaria", for example. There are more than 4,800 programs of study registered articles on treatments and vaccines. We know more about SARS-CoV-2 and COVID-19 than about other diseases that we have been studying for decades.
You can consult:
The Medical Letter on Drugs and Therapeutics (Treatments considered for COVID-19).
COVID-19 Global literature on coronavirus disease, WHO.
COVID-19 Vaccine & Therapeutics Tracker.
More than 200 new vaccines.
A year ago, eight new SARSCoV-2 coronavirus vaccine projects were highlighted. According to portal bioRENDER there are more than 195 candidates, at least 71 already in clinical trials, using all subject technologies: live attenuated viruses, inactivated viruses, protein subunits, recombinant viral vectors, virus-like particles (VLP), DNA, RNAm, etc. Never before has so much money been invested and there has been so much partnership for the development of vaccines between public and private entities, centers of research, universities, pharmaceutical companies, companies, NGOs. Some projects have been abandoned (such as the proposal of Imperial College London/Morningside Ventures based on self-replicating RNA, those of Merck/ Themis Bioscience/Institut Pasteur and Merck/ IAVI based on recombinant viral vectors, or that of the Australian University of Queensland that combined proteins with adjuvants), but others have already been authorized by the WHO: Pfizer/BioNTech and Moderna with RNAm technology, AstraZeneca/Oxford and Sputnik V with recombinant adenovirus technology, and China's Sinopharma, with inactivated coronaviruses. At least 20 other vaccines are already in Phase III clinical trials and may be approved in the coming weeks/months, if the results are satisfactory.
You can consult:
Draft landscape and tracker of COVID-19 candidate vaccines.
Challenges in ensuring global access to COVID-19 vaccines: production, affordability, allocation, and deployment. The Lancet, February 12, 2021.
Coronavirus Vaccine Tracker (New York Times).
3. RNAm vaccines are very safe.
One of the possible serious effects of vaccines is anaphylaxis, a life-threatening allergic reaction that usually occurs soon after the vaccine is administered. An analysis has been made of data from the first month of vaccination in the U.S., where more than 17.5 million doses have been administered (exactly 9,943,247 of the Pfizer/BioNTech vaccine and 7,581,429 of the Moderna vaccine). The Vaccine Adverse Event Reporting System (VAERS) has recorded only 66 cases of anaphylaxis (47 with the Pfizer/BioNTech vaccine and 19 with the Moderna vaccine). This is less than 4 cases per million doses or 0.0003% of all doses tested. Twenty-one (32%) of those 66 cases had had previous cases of anaphylaxis for other reasons. No deaths were detected. When compared with the issue of COVID-19 cases, the sequelae of the disease and the issue of deaths, the benefit of the vaccines greatly outweighs the possible adverse effects, and allows us to state that, for the time being, mRNA vaccines are very safe.
You can consult:
Reports of Anaphylaxis After Receipt of mRNA COVID-19 Vaccines in the US-December 14, 2020-January 18, 2021. JAMA, February 12, 2021.
4. Vaccines are effective.
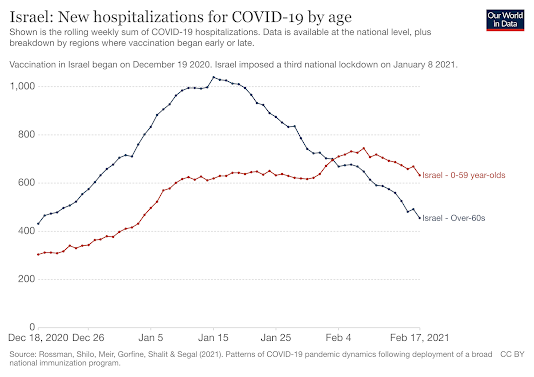
Israel is the country with the largest vaccinated population. As of early February, more than 3.67 million Israelis had received the first dose of the Pfizer/BioNTech mRNA vaccine since the campaign began in December. This represented about 40% of the country's population. More than 28% had also received the second dose. Among those over 60, more than 80% had been vaccinated. Preliminary data show that vaccination is proving effective. group The issue of infections is decreasing significantly, especially among those over 60. In this age group, there have been 56% fewer infections and 42% fewer hospitalizations and 35% fewer deaths from COVID-19 after the second dose. The results with the two doses are excellent: of the 523,000 Israelis vaccinated with two doses there are only 544 cases of COVID-19, only 4 cases of severe COVID-19 and zero deaths. These data confirm those obtained in previous clinical trials.
But we do not have to go all the way to Israel; in Asturias, on February 15, the number of people killed by COVID-19 since the beginning of the pandemic had surpassed the figure of 2,000. Among them, there was a large proportion of people living in nursing homes, where the impact has been very considerable. However, status is now beginning to be relatively under control thanks to vaccination efforts specifically targeting residents and caregivers. The effect of the vaccine is evident when comparing mortality among elderly people living in nursing homes (almost all of whom are vaccinated), where it falls sharply, and the issue number of deaths among people living outside the homes (not vaccinated), where it increases considerably.
Not only that, but the results of a preliminary study in England have just been published showing that the Pfizer/BioNTech mRNA vaccine is effective in preventing infection in symptomatic and asymptomatic adults, even against the "British" B1.1.7 variant.
You can consult:
Patterns of COVID-19 pandemic dynamics following deployment of a broad national immunization program, Februery 9, 2021.
Vaccinations and the impact of COVID-19 - our continuously-updated data for Israel (Our Word in Data).
Mortality with coronavirus in Asturias, 2020 report #29: 17.02.2021.
Effectiveness of BNT162b2 mRNA Vaccine Against Infection and COVID-19 Vaccine Coverage in Healthcare Workers in England, Multicentre Prospective Cohort Study (the SIREN Study). The Lancet, February 22, 2021.
5. Confidence in vaccines increases.
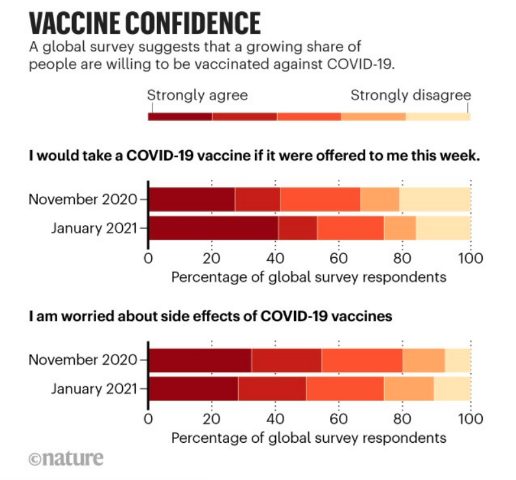
After more than 160 million doses of COVID-19 vaccines have been administered, public confidence in the vaccines is increasing. For example, a survey survey of 13,500 people in 15 countries in Europe, Asia and Australia was conducted between November 2020 and January 2021. In the month of November, before countries began approve the vaccines, only about 40% of respondents would take the COVID-19 vaccine and more than 50% were concerned about possible side effects. By January, more than half would get the vaccine and the issue of those concerned about side effects had decreased slightly.
The United Kingdom was the country where most people said they were willing to be vaccinated (up to 78% of those surveyed) and in Spain the proportion of people willing to be vaccinated rose from 28% in November to 52% in mid-January.
You can consult:
Trust in COVID vaccines is growing. Nature, February 10, 2021.
6. The immune response against the virus lasts at least eight months.
Serological tests that measure antibodies to SARS-CoV-2 do not reflect the full potential, duration and report of the immune response to the virus. Knowing how long the immune response against the virus lasts is critical in determining protection against reinfection, disease severity, and vaccine efficacy. It has been shown that, although there is some heterogeneity in the response according to each individual, most of the people tested maintain a robust humoral (antibodies) and cellular (T-lymphocytes) immune response up to 6-8 months after infection, regardless of whether they are mild or severe.
You can consult:
Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science, February 5, 2021.
Persistence of SARS-CoV-2 specific B- and T-cell responses in convalescent COVID-19 patients 6-8 months after the infection. Cell, February 3, 2021.
7. New treatments for the most severe cases.
We already know that COVID-19 is much more than pneumonia. Much more is known about the disease and, although we do not yet have a specific antiviral that inhibits the virus, there are combinations of treatments that greatly improve the prognosis and reduce mortality in the most severe cases: antivirals, anti-inflammatory drugs, anticoagulants, corticosteroids, cytokine storm inhibitors, monoclonal antibodies, ... There are more than 400 ongoing clinical trials inwhich different treatments and combinations are being tested. For example, according to the international clinical essay Recovery, the combination of tocilizumab (a monoclonal antibody directed against the interleukin-6 receptor, C for the treatment of rheumatoid arthritis) and dexamethasone (a potent synthetic glucocorticoid that acts as an anti-inflammatory and immunosuppressant), can reduce deaths in the most severe patients with COVID-19 by almost half.
On the other hand, preventive treatment with anticoagulants in hospitalized COVID-19 patients is associated with 30% lower 30-day mortality and no adverse bleeding effects.
You can consult:
RECOVERY trial shows tocilizumab reduces deaths in patients hospitalised with COVID-19, NIHR, February 11, 2021.
Early initiation of prophylactic anticoagulation for prevention of coronavirus disease 2019 mortality in patients admitted to hospital in the United States: cohort study, BMJ, February 11, 2021.
COVID-19 Research Registry - Treatment (ASM).
8. No flu
There was serious concern about how the overlap of SARS-CoV-2 with other respiratory pathogens prevalent in the winter months would behave. A "perfect storm" status in which SARS-CoV-2 coincided with other viruses, such as influenza or respiratory syncytial virus, which cause bronchiolitis and pneumonia and are responsible for frequent hospitalizations and deaths in certain sectors of the most vulnerable population, could not be ruled out. It had been suggested that the risk of death in persons infected by influenza and SARS-CoV-2 simultaneously was higher than in those infected only by the coronavirus, especially in those over 70 years of age. The coincidence of several respiratory viruses with SARS-CoV-2 could have caused real carnage in the elderly.
The good news is that this season the flu and other respiratory viruses have disappeared, both in the months of June-August in the southern hemisphere and now in the northern hemisphere. We cannot rule out that this could perhaps pose a problem next year (seasons in which influenza causes higher mortality are usually preceded by milder seasons), but this year has been a real relief to health systems.
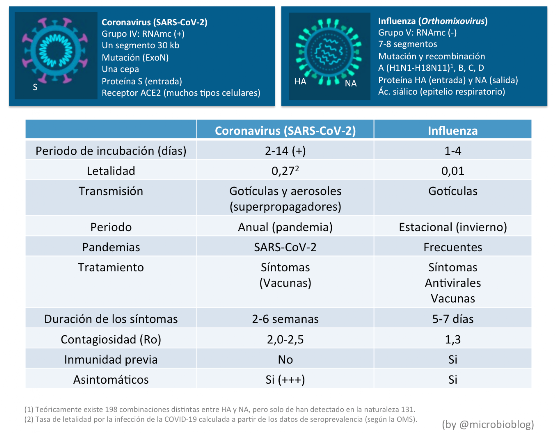
Several causes may explain this decline in influenza. First, it should be remembered that SARS-CoV-2 and the influenza virus are very different viruses.
It is very likely that the shorter incubation period of influenza, the existence of previous immunity, the intense vaccination campaign of this year, the measures of confinement, reduction of travel, use of masks, hygiene, social distancing, etc. have had a greater effect in reducing the transmission of this virus. On the contrary, the transmission of the coronavirus is also much more influenced by the effect of aerosols, the role of superpropagators and asymptomatic people.
9. We can follow the evolution of the virus in real time.
The effect that new genetic variants of SARS-CoV-2 may have on vaccination and the course of the pandemic is an uncertainty. Because genetic changes can potentially have an effect on how the virus behaves, their analysis and monitoring is critical. The good news is that we now have the ability to track the real-time evolution of the virus and the emergence of new genetic variants. There are already more than 260,000 SARS-CoV-2 genome sequences available at instructions at data. These sequences come from as many isolates obtained from human samples from February last year to the present. Although nucleotide changes are the primary source of variation Genetics of SARS-CoV-2, insertions, deletions and even recombinations have also been detected.
All this allows for phylogenies (relationships of "kinship" between viral variants) that can be used to make temporal estimates (when new variants emerge), characterize how the virus spreads geographically, reconstruct the epidemiological dynamics within a region and analyze how they adapt over time. data The analysis of SARS-CoV-2 sequences is unprecedented, with more than 580,000 shared sequences at database GISAID (Global Initiative on Sharing Avian Influenza Data). This is the first time that the evolution of a pandemic virus is being tracked in real time.
You can consult:
Insights from SARS-CoV-2 sequences. Science, January 29, 2021.
SARS-CoV-2 Proteome-3D Analysis (University of Cambridge).
CoVariants (Institute of Social and Preventive Medicine, University of Bern).
10. The global pandemic is decreasing.
We do not know how the pandemic will develop in the coming months. Given the intensity it has had so far, it is likely that there will be new waves, but perhaps of lesser intensity. We do not know what a possible fourth wave will be like, nor the effect that the new genetic variants that are appearing may have, but the good news is that at a global level the pandemic is decreasing at this time.
(source)
Perhaps it is a combination of several factors: the virus behaves seasonally, the population is acquiring some immunity from group by natural infection or vaccines, perhaps the virus in this natural process of variation and mutation is drifting to less virulent forms and is adapting to its new host. We do not know for sure, but for the time being there is still reason for hope.